Established and emerging biomarkers in immuno-oncology: New Insights from the UK Biobank Pharma Proteomics Project
- Inflammation , Oncology , Protein biomarkers
- Read time: 9 minutes
Immuno-oncology mouse models are indispensable tools for elucidating the immune response to cancer and the dynamics within the tumor microenvironment (TME). In this context, the multiplex measurement of immune mediators is critical to identifying key proteins and pathways involved in the anti-tumor response.
There are different approaches to deciding which analytes to analyze in order to get a comprehensive profiling of the tumor microenvironment (TME) or to identify relevant biomarkers in mouse models. One approach that has recently become available comes from the UK Biobank Pharma Proteomics Project (UKB-PPP). Utilizing Olink® technology, this project analyzed over 50,000 samples to integrate protein measurements with longitudinal healthcare data and estimate the impact of 3,000 proteins on future disease risks. This extensive data library is now accessible through Olink Insight, a free and user-friendly web platform designed for non-data scientists here: https://insight.olink.com/.
Drawing from the data derived from this analysis, this blog post discusses selected immune-related proteins that demonstrated statistically significant associations with the cancer types included in the UKB-PPP study. This encompasses both well-established biomarkers and some emerging, yet highly promising, candidates.
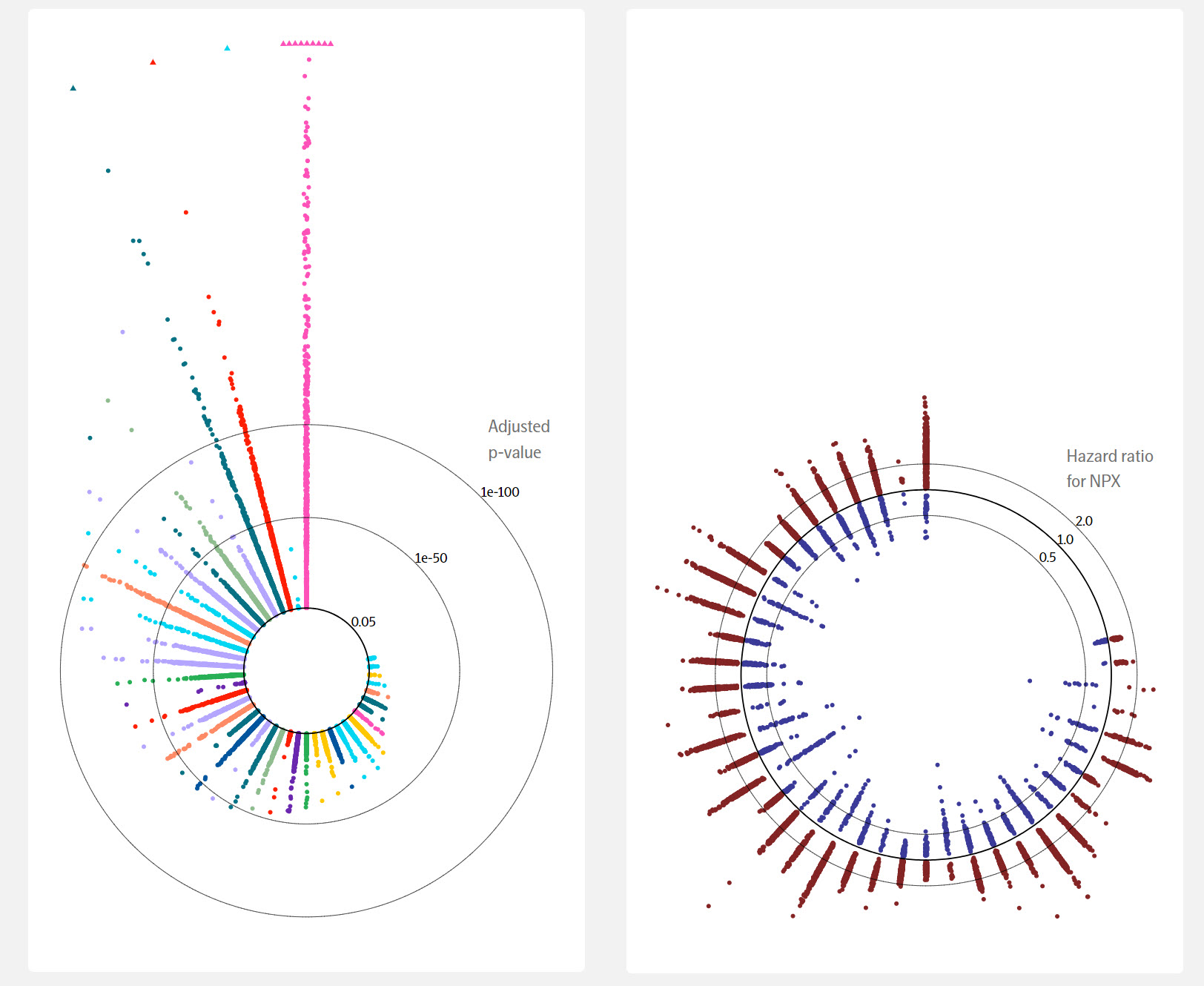
Figure 1: The UKB proteomics data was linked to hospital records and other healthcare data included in UKB. The risk associations were analyzed for 106 diseases that were selected from the PheWAS phenotype ontology. A requirement was implemented for 150 incident cases over a 10-year period from entering the UKB program, excluding individuals with a prior diagnosis of the disease. P-values were extracted as measures of protein-disease associations and adjusted for multiple testing according to the Benjamini-Hochberg method. Cox regression was employed to estimate the effect of protein levels on subsequent risk of developing disease. In this figure, circular visualizations, both the adjusted p-value (left) and the HR (right) are presented for each statistically significant protein in each focus disease, including cancers that are mentioned in this blog post.
Essential biomarkers in Immuno-oncology research
Perhaps unsurprisingly, UKB-PPP uncovered significant associations between cancer risk and pro-inflammatory cytokines. These are known to play dual roles in cancer, supporting both tumor growth and anti-tumor immunity, depending on the environment and concentration levels. A significant association of the following cytokines with risk of cancer was observed:
- IFN-γ was significantly associated with risk of cancer of prostate (HR = 0.92, p = 0.036*)
- IL-1β was significantly associated with the risk of cancer of bronchus lung (HR=1.2, p=0.019*)
- IL-6 was significantly associated with the risk of cancer of bronchus lung (HR=1.2, p= 0.0000160*)
Immune checkpoints are also highly relevant and play well established roles in the TME. Cancer cells evolve to evade these checkpoints and, therefore, these are generally promising targets of therapies (e.g., nivolumab, ipilimumab). Unsurprisingly, also PD-L1 is significantly associated with risk of cancer of bronchus lung (HR = 1.6, p = 0.00011*).
There is a fundamental importance of growth factors in regulating key cellular processes involved in cancer development and progression, as well as in potential therapeutic resistance. For instance, M-CSF had a strong risk association with cancer of bronchus lung and cancer of kidney and renal pelvis (HR = 1.9, p = 7.22e-5 and HR = 2.9, p = 0.00011, respectively)
HGF and its receptor c-MET hyperactivation is linked to malignant cancers. Although its expression is generally associated with poor outcomes and therapeutic resistance, there is evidence suggesting it also induces anti-tumor immunity. HGF was shown to be associated with risk of cancer of bronchus lung (HR = 1.1, p = 0.0014*)1
Emerging and promising biomarkers in I-O research
Beyond these well-established markers, the UKB-PPP analysis unveiled markers that are more rare in common panels, but highly promising, as described below:
CCL chemokines play a crucial role in recruiting and regulating different immunosuppressive cell populations within the TME, favoring tumor growth, invasion, and metastasis. Specifically, CCL22 and CCL17 promote an immunosuppressive tumor environment by recruiting Tregs, which can inhibit anti-tumor immune responses and promote tumor growth and metastasis. The presence of CCL22 in the TME has been associated with poor prognosis in various cancers2. Apart from supporting an immunosuppressive environment like the other CCL chemokines, CCL11, or Eotaxin, facilitates proliferation, migration and invasion of cancer cells across various cancer types. In part due to their role in conferring resistance for apoptosis in cancer cells and contributing to tumor angiogenesis2. The UKB-PPP disease risk data revealed that CCL22, CCL17 and CCL11 are significantly associated with cancer of bronchus lung (HR = 1.3 | p = 7.57e-6, HR = 1.2 | p = 0.0024, HR = 1.4 | p = 0.011, respectively*).
CXC chemokines are generally upregulated in cancer patients. Many of them associate with tumor progression, cancer metastasis and chemo-resistance via promotion of angiogenesis3. CXCL9 levels associated with cancer of bronchus lung (HR = 1.3, p = 7.57e-6*) and CX3CL1 with other non-epithelial cancer of skin (HR = 1.3, p = 0.0027*).
FGF-21 is a metabolic regulator highly expressed in hepatic and pancreatic cells involved in tumor cell proliferation, apoptosis, migration, and invasiveness, with reports of its involvement in prostate and endometrial cancer. FGF-21 may represent a therapeutic opportunity for cancers associated with metabolic abnormalities. In the UKB-PPP data, FGF-21 showed a strong association with risk for cancer of bronchus lung (HR = 2.0, p = 5.63e-8*)4,5.
IL-22 is emerging as a significant player in the complex interplay between inflammation, immunity, and cancer. IL-22 is crucial for tissue repair and has been associated with the progression and development of several types of cancer, including those of the lung, breast, gastric, pancreatic, and colon6. In the tumor microenvironment, IL-22 has a dual role. It is capable of both promoting tissue healing and contributing to tumor development. Understanding the precise mechanisms through which IL-22 influences cancer progression is crucial for developing strategies to harness or inhibit its effects within the tumor microenvironment6. According to the disease risk study, IL-22 is significantly associated with cancer of bronchus lung (HR = 1.3, p = 1.58e-5*).
IL-9 has traditionally been recognized for its involvement in allergic diseases and responses to parasite infections. The exploration of IL-9’s role within the context of cancer has recently gained momentum. Studies are uncovering its potential anti-tumor effects, marking IL-9 as an increasingly attractive cytokine for investigation7. In line with that, the UKB-PPP data has shown that IL-9 is significantly associated with cancer of bronchus lung (HR = 1.2, p = 0.040*)
The UKB-PPP is an invaluable resource for guiding the selection of biomarkers in immuno-oncology and other diseases. However, it is important to consider that immune mediators also hold promise in various aspects of immuno-oncology research beyond the risk of developing cancer. These include understanding responses to immunotherapy and assessing the risk of immune-related adverse events or cytokine release syndrome. In this context, established markers like IL-2, IL-17A, and GM-CSF are also noteworthy.
A unique panel bridging essential and emerging biomarkers
Integrating all the immune mediators discussed, plus some additional ones, into a single panel provides a comprehensive view of the tumor microenvironment (TME) and immune response to cancer in preclinical mouse models. Olink has carefully designed a unique multiplex panel. This panel incorporates 43 highly promising immune-related proteins consisting of essential and emerging biomarkers. Remarkably, the absolute quantification of these 43 analytes can achieved from just 1 µL, conserving precious samples. This efficiency allows for the execution of studies that were previously unthinkable, including longitudinal monitoring of immune responses and detailed profiling of highly limited sample types.
For further details, including the complete assay list and selected validation data, please fill out the form below.
*The estimated NPX hazard ratios (HRs) and their associated p-values were extracted as measures of protein-disease associations. Adjusted p < 0.05 means a statistically significant association, while HR presents the effect size. For more information on the data analysis of the UKB-PPP, refer to the Data and methods section of the Insight Data story on disease risk https://insight.olink.com/data-stories-articles/ukb-diseases-univariate/#section-data-and-methods.
References:
- Papaccio F, Della Corte CM, Viscardi G, Di Liello R, Esposito G, Sparano F, Ciardiello F, Morgillo F. HGF/MET and the Immune System: Relevance for Cancer Immunotherapy. Int J Mol Sci. 2018 Nov 14;19(11):3595. doi: 10.3390/ijms19113595. PMID: 30441809; PMCID: PMC6274701.
- Korbecki, J.; Kojder, K.; Simińska, D.; Bohatyrewicz, R.; Gutowska, I.; Chlubek, D.; Baranowska-Bosiacka, I. CC Chemokines in a Tumor: A Review of Pro-Cancer and Anti-Cancer Properties of the Ligands of Receptors CCR1, CCR2, CCR3, and CCR4. Int. J. Mol. Sci. 2020;21(21):8412. doi: 10.3390/ijms21218412.
- Wu T, Yang W, Sun A, Wei Z, Lin Q. The Role of CXC Chemokines in Cancer Progression. Cancers (Basel) 2022 Dec 28;15(1):167. doi: 10.3390/cancers15010167. PMID: 36612163; PMCID: PMC9818145.
- Lu W, Li X, Luo Y. FGF21 in obesity and cancer: New insights. Cancer Lett 2021 Feb 28;499:5-13. doi: 10.1016/j.canlet.2020.11.026. PMID: 33264641; PMCID: PMC7779663.
- Jagodzińska A, Chudecka-Głaz A, Michalczyk K, Pius-Sadowska E, Wieder-Huszla S, Jurczak A, Machaliński B. The Diagnostic Role of FGF 21 in Endometrial Cancer and Other Pathologies of the Uterine Corpus. Diagnostics (Basel) 2023 Jan 22;13(3):399. doi: 10.3390/diagnostics13030399. PMID: 36766504; PMCID: PMC9914808.
- Papaccio F, Della Corte CM, Viscardi G, Di Liello R, Esposito G, Sparano F, Ciardiello F, Morgillo F. HGF/MET and the Immune System: Relevance for Cancer Immunotherapy. Int J Mol Sci. 2018 Nov 14;19(11):3595. doi: 10.3390/ijms19113595. PMID: 30441809; PMCID: PMC6274701.
- Vinokurova, D.; Apetoh, L. The Emerging Role of IL-9 in the Anticancer Effects of Anti-PD-1 Therapy. Biomolecules. 2023;13:670. doi: 10.3390/biom13040670.